Quick Details
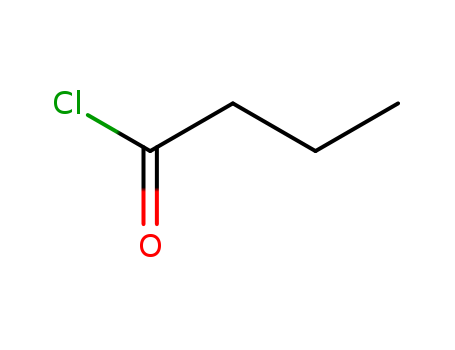
- ProName: Butyryl chloride
- CasNo: 141-75-3
- Molecular Formula: C4H7ClO
- LimitNum: 0
Superiority
FORMULA CH3CH2CH2COC1
MOL WT. 106.55
PHYSICAL AND CHEMICAL PROPERTIES
PHYSICAL STATE colorless to light yellow liquid with a str…
Details
FORMULA CH3CH2CH2COC1
MOL WT. 106.55
PHYSICAL AND CHEMICAL PROPERTIES
PHYSICAL STATE colorless to light yellow liquid with a strong pungent odor.
MELTING POINT -89
BOILING POINT 102
SPECIFIC GRAVITY 1.026
VAPOR DENSITY 3.67
REFRACTIVE INDEX 1.4120
NFPA RATINGS Health 3, Flammability 3, Reactivity: 0
FLASH POINT 18
STABILITY It undergoes vigorous hydrolysis with water.
GENERAL DESCRIPTION & APPLICATIONS
Acyl is a radical formed from an organic acid by removal of a hydroxyl group. The general formula of acyl compound is RCO-. hcylation reaction involves substitution by an electron donor (nucleophile) at the electrophilic carbonyl group (C=O). The carboxytic acid (X = OH) itself can function as an acylating agent when it is protonated by a strong acid catalyst as in the direct esterification of an alcohol. Two common acylation agents, with the general formula RCOX, are acid halides (X = halogen atom) and anhydrides (X = OCOR). It is used in manufacturing peroxides such as a benzoyl peroxide and t-butyl perbenzoate. It is also used in the synthesis of benzophenone and its derivatives used in manufacturing pesticides, pharmaceuticals, perfume fixative, polymerization catalyst, benzolating agents, and dyestuffs, n-Butyryl Chloride, C-4 Acyl halide, is a colorless to light yellow liquid with a strong pungent odor; decomposes in water; soluble in almost aprotic organic solvents. It is used as an intermediate for organic synthesis for the preparation of pharmaceuticals, agrochemicals, dyes, cellulose esters, and peroxide compounds.