Superiority
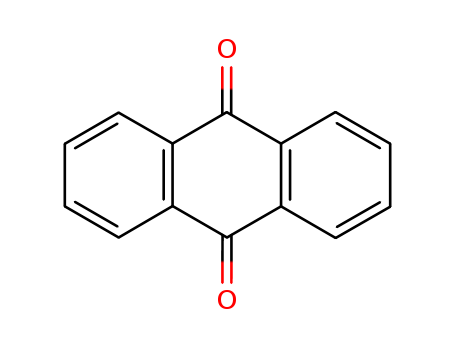
FORMULA :C14H802
MOL WT. 208. 22
H S. CODE 2914. 61
TOXICITY Oral rat LD50: 15 gr/kg
PHYSICAL AND CHEMICAL PROPERTIES
PHYSICAL STATE yellow green…
Details
FORMULA :C14H802
MOL WT. 208. 22
H S. CODE 2914. 61
TOXICITY Oral rat LD50: 15 gr/kg
PHYSICAL AND CHEMICAL PROPERTIES
PHYSICAL STATE yellow green solid
MELTING POINT: 284 - 286
BOILING POINT: 379 - 381
SPECIFIC GRAVITY 1.44
SOLUBILITY IN WATER Insoluble
AUTOZINITION: 650
VAPOR DENSITY 7.16
NFPA RATINGS Health:O Flammability: i Reactivity:
FLASH POINT: 185
SfABILITY Stable under ordinary conditions
GENERAL DESCRIPTION & APPLICATIONS
Anthraquinone, a polycyclic aromatic hydrocarbon containing two opposite carbonyl groups (C=O) at 9,10 position, is yellow or light gray to gray-green crystal powder; insoluble in water. , In nature, it occurs in plants (aloe, cascara sagrada, senna, and rhubarb), fungi, lichens, and insects as a parent material for coloring of yellow, orange, red, red-brown, or violet. It is commercially produced by several ways including by oxidation of anthracene with chromic acid, the condensation of benzene and phthalic anhydride, followed by dehydration for cyclization, and diene Diels-Alder reaction. Anthraquinone is the most important quinone derivative of anthracene as the parent substance of a large class of dyes and pigments. One of the oldest mordant dye, alizarin, is the anthraquinone derivative. Anthraquinone is a starting material for the production of coloring compounds, antioxidants, and polymerization inhibitors. Its derivatives are widely used as intermediates for dyes, pigments, photographic chemicals, and paints. Anthraquinone is used in paper industry as a catalyst to increase the pulp production yield and to improves the fiber strength through reduction reaction of cellulose to earboxylic acid. Natural anthraquinones have cathartic properties. Anthraquinones derivatives are also used in manufacturing drugs. Mitoxantrone, an antineoplastic is an example.